Epoxy coatings are some of the most widely used materials in industrial and marine environments. Epoxy materials typically have excellent adhesion, chemical resistance and water resistance, which make them first-rate candidate coatings/linings for these environments. Epoxies are formulated as two component materials, with one component housing an epoxy resin and the other component containing a curing agent. Curing agents are used to react with the epoxy resin to create a cross-linked coating resin. In fact, many epoxy materials have what is known as an “induction time,” or “sweat-in time,” where the coating is mixed and allowed to react for a period of time (e.g., 30 minutes) so that cross-linking of the components can initiate prior to application of the product. Though epoxy materials can have many co-reactive curing agents, the focus of this article is on amine cured epoxies.
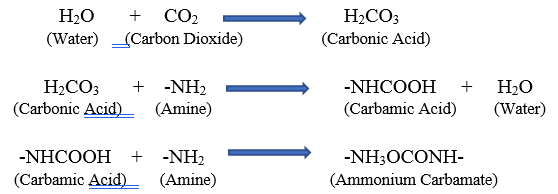
Amine cured epoxies have an active amine functional group that react with the epoxy functional groups in the resin component. Amine exudate (blush) is a chemical reaction that occurs between water, carbon dioxide, and the amine component of epoxy coatings. Amines are hygroscopic, which means that they tend to absorb moisture from the air and are prone to react with available moisture and carbon dioxide. The chemical reactions that form the exudate are illustrated below:
From this point, the carbamate continues to react with carbon dioxide and forms ammonium bicarbonate. This chain of reactions can lead to a sticky, oily, or waxy film appearing on the surface of the cured epoxy material, which is largely due to the ammonium bicarbonate salts. If the surface is to be overcoated, the presence of amine blush can act as a bond breaker between the coating layers. The adhesion of the two layers will be severely decreased and will likely lead to coating failure.
Aside from the serious issue of decreased adhesion of subsequent coats, amine blush can cause a myriad of other difficulties when working with epoxies, including aesthetics, an increased tendency to yellow, decreased stain resistance, lower gloss retention, softening of the coating, reduced chemical resistance, or other altered characteristics. In addition, when the amine component reacts with water and carbon dioxide, it can leave some of the epoxy resin unreacted. This leaves the coating in an under-cured state and the resistance properties of the coating may be compromised.
While some types of epoxies may be more susceptible to blush, even those that may not have this same susceptibility can fall victim to this reaction if the conditions are present. For this reason, it is important to minimize the contributing factors. The best way to achieve this is to control exposure of amine materials to moisture and carbon dioxide, and to ensure that the coating reacts the way the manufacturer intended.
Firstly, it is important to understand the role that temperature plays during the curing of materials. Applying a coating in low temperature slows the rate of reaction and increases the likelihood of blush because of longer amine exposure. Additionally, it is always important to be aware of the dew point temperature (the temperature that moisture will condense). Coating application is not advised when the temperature is below the dew point, and in the case of epoxies, if the temperature is hovering near the dew point, a drop in the temperature can result in condensation on the substrate or even on the coating. If low temperatures cause a problem during application, heat can be used to elevate the air and surface temperatures. This may present another challenge however, since the use of some heaters can create excess carbon dioxide and moisture (especially in spaces that are not well ventilated), which may dictate the use of dehumidification equipment. A case study where ideal temperature and humidity conditions were present, but poor ventilation practices resulting in amine blush is detailed in an article published in the April 2009 edition of The Journal of Protective Coatings and Linings (JPCL), “F-Files: Mechanisms of Failure The case of the yellowed water tank lining.”
As we’ve discussed, moisture in the air in combination with the amine component and carbon dioxide react to form an amine exudate or blush. High humidity can also result in blush formation. The dew point temperature plays an important role when high humidity conditions are present, and it is important to verify the surface temperature is at least 5°F above dew point to avoid condensation during the curing process. And needless to say, application during any sort of rain event over a visibly wet surface would increase the opportunity for the amine to react with moisture.
To further reduce the risk of amine blush, it is imperative that the components of the coating react in the way which they were formulated to react. For example, if the induction time is neglected and the product is applied immediately after mixing, the amine will not have had adequate time to react with the epoxy component but will be available to react with moisture in the air, which can lead to amine blush. The mix ratio of the components is equally important. If an excess quantity of the amine component is added during mixing, the epoxy component will react with the amine as appropriate, and the balance will remain as free unreacted amine until it comes into contact with moisture and carbon dioxide.
There are several ways to detect whether amine blush is present, or to verify its presence if it is suspected to have occurred. As described earlier, amine blush typically manifests itself as an oily, or waxy film on the coating surface and is usually visible to the unaided eye. It often has a yellow, hazy, or milky appearance. The tacky nature of the film (once it begins to oxidize) can lead to dirt or debris pick-up. There are also field tests that can be performed. One such test is performed using pH paper. Amine blush is alkaline, so an elevated pH reading may indicate the presence of an amine exudate. A limitation to pH testing is that other factors like surface contamination may trigger a high pH, but is not due to the presence of an amine blush. In addition to pH testing, a field test kit can be used to detect the presence of carbamates on the coating surface, compared to a clean, control surface.
If a coating failure has occurred, Fourier transform infrared spectroscopy (FTIR) can be used to provide forensic evidence of its presence; however FTIR is not a quantitative technique and the amount of amine exudate sufficient to cause delamination has not been quantified.
If an amine blush has formed on the surface it is not too late to correct the problem. The coating manufacturer should always be consulted before attempting to recoat. In fact, depending on the formulation of the product, the manufacturer may expect amine blush to occur under cool damp, or high humidity conditions. Since amine blush is water-soluble, its removal is not inherently complicated. Oftentimes, the manufacturer will recommend washing the surface with detergent and water or a degreaser prior to overcoating. In these cases, it is important to verify the surface has been thoroughly rinsed and that all cleaners have been removed prior to recoating.
If the blush is present as more of a hazy top surface as opposed to a film, gentle abrasion of the surface may be a more effective removal process. However, if the blushed coating is not intended to be topcoated, care should be taken to not damage the texture, gloss or finish of the coating.
For more information contact Valerie Sherbondy: vsherbondy@kta.com


This was excellent reading. We are now finding similar issues with our intumescent fire proofing.
The second coat seems to have never bonded to the first coat and primer. Exposure to the environment let to cracking and once water ingressed behind it created total separation. We also detected an opaque waxy type substance on the surface between the coats where separation had occurred. This is currently being analyzed but could be amine blush.
Very nice article. Thanks Julia. We are facing similar blushing issue while new shipbuilding and painting of decks in Japan in low temperatures.